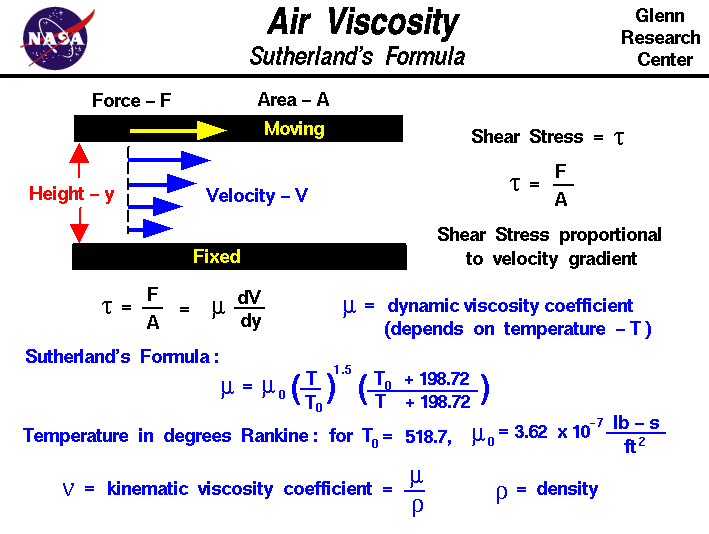
Specific heat capacity (C v) air at 0☌ and 1 bara: 0.7171 kJ/kgK = 0.17128 Btu(IT)/(lb m ☏) or kcal/(kg K).Specific heat capacity (C p) air at 0☌ and 1 bara: 1.006 kJ/kgK = 0.24028 Btu(IT)/(lb m ☏) or kcal/(kg K).Liquid density at boiling point and 1 bar: 875.50 kg/m 3 = 54.656 lb/ft 3.Bulk modulus elasticity: 1.01325 x 10 5 Pa or N/m 2.A scaling analysis based on Navier-Stokes equations is presented at the end, and the predicted θ(c) matches with experimental observations without any additional fitting parameters. Furthermore, we observed a viscous wetting regime only on surfaces with an equilibrium contact angle θ(eq) smaller than a critical angle θ(c) depending on viscosity. For relatively high viscosity liquids, the inertial wetting time increases with liquid viscosity, which may due to the viscous damping of the surface capillary waves. For low viscosity liquids, the duration of inertial wetting corresponds to the time of capillary wave propagation, which can be determined by Lamb's drop oscillation model for inviscid liquids. It was further found that surface wettability does not affect the duration of inertial wetting, whereas the viscosity of the liquid does. 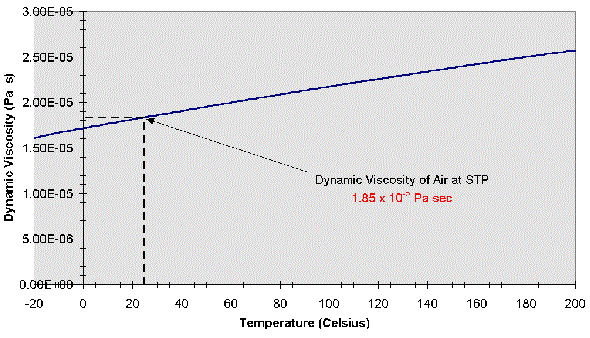
In contrast, the exponent of the power law does only depend on surface wettability as also reported in literature. In the early inertial wetting regime, the coefficient of the wetting power law increases with surface wettability but decreases with liquid viscosity. We show that surface wettability and liquid viscosity influence wetting dynamics and affect either the coefficient or the exponent of the power law that describes the growth of the wetting radius. Solid surfaces with different lyophilic and lyophobic coatings (equilibrium contact angle θ(eq) of 0°-112°) were used to study the effect of surface wettability. Drop of glycerol water mixtures and pure water that have comparable surface tensions (62.3-72.8 mN/m) but different viscosities (1.0-60.1 cP) were used. In this paper, we experimentally investigated the dynamic spreading of liquid drops on solid surfaces.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
